Atrial and ventricular pacing (AP/VP) have been related to a higher risk of clinical atrial fibrillation (AF). Subclinical AF (SCAF) is detected as atrial high rate episodes by cardiac pacing devices. We aimed to determine whether the percentage of AP/VP is related with the development of SCAF.
MethodsFrom February 2012 to September 2015 we recruited consecutive patients with dual chamber pacemakers and no history of AF. SCAF (atrial high rate episodes), clinical AF (electrocardiographically documented) and cardiovascular events were registered.
ResultsA total of 249 patients (57% men; 75±9 years) were included. After a mean follow-up of 33±11 months, 38.5% developed SCAF and 10.4% AF. Patients with AP ≥ 50% presented higher risk of SCAF (62.5% vs 32.3%; OR, 3.48; 95%CI, 1.93-6.4; P<.01) and AF (18.7% vs 8.6%; OR, 2.4; 95%CI, 1.05-5.52; P <.05). Patients with VP ≥ 50% had higher risk of SCAF (46.4% vs 31.6%; OR, 1.87; 95%CI, 1.10-3.24; P <.05) and AF (25.9% vs 9.7%; OR, 2.7; 95%CI, 1.13-7.72; P <.05). AP ≥ 50% was an independent predictor for SCAF (HR, 2.44; 95%CI, 1.20-4.97; P=.014).
ConclusionsAtrial and ventricular pacing are related with higher risk of developing subclinical and clinical AF in patients with dual-chamber pacemakers and no history of previous AF. Our data suggest, that patients presenting a high percentage of AP should be closely followed during routinely pacemaker check-ups assessing for SCAF.
La estimulación auricular y ventricular (AP/VP) se han relacionado con un mayor riesgo de fibrilación auricular (FA) clínica. La FA subclínica (SCAF) se puede detectar mediante marcapasos como episodios de frecuencia rápida auricular. El objetivo fue determinar si el porcentaje de AP/VP se relacionaba con el desarrollo de SCAF.
MétodosDesde febrero 2012 hasta septiembre 2015 se incluyeron pacientes consecutivos con marcapasos bicamerales y sin historia de FA. Se registraron episodios de SCAF (episodios de frecuencia rápida auricular), FA clínica (documentada electrocardiográficamente) y eventos cardiovasculares.
ResultadosSe incluyeron 249 pacientes (57% varones; 75 ± 9 años). El seguimiento medio fueron 33 ± 11 meses; el 38,5% desarrolló SCAF y el 10,4% FA. Los pacientes con AP ≥ 50% presentaban mayor riesgo de SCAF (62,5 frente a 32,3%; OR = 3,48; IC95%: 1,93-6,4; p < 0,01) y FA (18,7 frente a 8,6%; OR = 2,4; IC95%: 1,05-5,52; p < 0,05). Los pacientes con VP ≥ 50% tenían más riesgo de SCAF (46,4 frente a 31,6%; OR = 1,87; IC95%: 1,10-3,24; p < 0,05) y FA (25,9 frente a 9,7%; OR = 2,7; IC95%: 1,13-7,72; p < 0,05). AP ≥ 50% era predictor independiente de SCAF (HR = 2,44; IC95%: 1,20-4,97; p = 0,014).
ConclusionesLa estimulación auricular y ventricular se relaciona con mayor riesgo de desarrollar SCAF y FA en pacientes con marcapasos bicamerales sin historia de FA. Esto sugiere que los pacientes con mayores porcentajes de AP deben ser vigilados cuidadosamente en los chequeos rutinarios de marcapasos, descartando SCAF.
Atrial fibrillation (AF) is the most common sustained arrhythmia and it has been related to increased morbidity and mortality.1,2 Nowadays, there is a growing interest both in its early detection and in a better discrimination of those patients at a higher risk that would obtain more benefit from anticoagulation to prevent thromboembolic complications. Dual-chamber pacemakers have demonstrated to provide reliable detection of subclinical AF (SCAF), defined as atrial high rate episodes (AHRE), and clinical AF (electrocardiographically [ECG] documented).3 AHRE have been related to a higher risk of both clinical AF and thromboembolic events.4 Previous studies have related atrial pacing (AP) with a reduction in AF recurrences5; however, this relationship is controversial, since other authors suggest an increased risk of AF related to AP.6 Ventricular pacing (VP) has also been related to an increased risk of AF due to different mechanisms.7 Therefore, pacing is related to an increased risk for future clinical AF development.
The aim of this study was to identify if AP and/or VP are related not only to higher rates of clinical AF but also to SCAF, as a previous stage in the natural course of AF, in patients with dual-chamber pacemaker and no prior history of this arrhythmia.
MethodsPatient populationPatients in sinus rhythm, without a history of previous episodes of AF and with a St. Jude Medical dual-chamber pacemaker, were prospectively included in the study. Baseline patients’ characteristics including cardiovascular risk factors, prior heart disease, indications for pacemaker implantation, and medications were recorded. Subclinical AF (SCAF) was defined as AHRE. Clinical AF was defined as ECG documented AF. Left ventricular dysfunction was defined as left ventricular ejection fraction <50%. Chronic kidney disease was defined as glomerular filtration <60 mL/min/1.73m2. Previous cardiomyopathy was defined as any previous structural heart disease.
The study protocol was presented and approved by the Ethics Committee of our institution, and conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in the priori approval by the human research committee of the institution. Patients were recruited after implantation or in the scheduled follow-up visits from February 2012 onward. At inclusion, patients were informed of the investigational purpose of the study, and informed consent was obtained. Scheduled pacemaker checkups were planned at 3 months after inclusion and every year thereafter. The checkups were in person. All the pacemaker check-up data were downloaded in the patientś medical histories. No special surveillance was protocoled in patients presenting AHRE during follow-up.
Device settingsIt is the protocol of our arrhythmia unit to avoid right VP in all the dual-chamber pacemakers prolonging the atrioventricular interval and using other dedicated algorithms when necessary. The maximum extent of atrioventricular delay was up to 400 ms. All pacemakers were able to record and collect the electrogram on any AHRE episodes. For the purpose of this investigation, AHRE or SCAF were defined as episodes of atrial rate ≥ 225 beats per minute with a minimum duration of 5minutes or more. The episodes were identified and confirmed by an expert during the checkups. Intracavitary electrograms acquisition was programmed to confirm AF based on atrial electrograms during the episodes. Clinical AF was defined as ECG-documented AF. Episodes were confirmed by 2 experts on pacemaker checkups and electrogram interpretation.
Data analysisWe analyzed prospectively the incidence of SCAF (as AHRE) and clinical AF (ECG-documented) in patients in sinus rhythm without history of AF with dual-chamber cardiac devices and the percentages of AP and VP.
From February 2012, all the subsequent scheduled pacemaker check-ups were reviewed. Pacemaker atrial stimulation/sense parameters, atrial lead impedance, percentages of AP and VP, AHRE, automatic mode switch episodes, and noise episodes were recorded. The percentage of AP and VP was determined as the mean AP and VP, respectively, during the first three visits (that is 3 months after inclusion and then every year). Required adjustments during checkups were made following the routine protocols of our arrhythmia unit. Patients were classified as SCAF or as AF when one episode meeting the above described criteria was detected. Clinical AF was defined as ECG-documented. These episodes were documented in face-to-face follow-up visits and also through the electronic medical history.
A clinical evaluation was also performed including symptoms, hospital/emergency department admissions, changes in treatment (antiarrhythmic and antithrombotic treatment), ECG documentation of AF, and mortality.
Cardiovascular events were defined as hospitalization for heart failure, AF and cerebrovascular events.
Statistical analysisQuantitative variables are expressed as the mean±standard deviation because they followed a normal distribution. Categorical variables are expressed as frequencies and percentages. Categorical variables were compared using the Chi-square test, and quantitative variables were evaluated using the Mann-Whitney U test. P values of <.05 were considered statistically significant.
The following variables were included in the univariate Cox regression model: age, hypertension, diabetes, tobacco use (smoking status), previous cardiomyopathy, ischemic cardiomyopathy, valvular prostheses, left ventricular dysfunction, hospitalization for heart failure, history of cerebrovascular accident, renal function, CHA2DS2-VASc, treatment with angiotensin-converting-enzyme inhibitors/angiotensin II receptor blockers, beta-blockers, statins, aspirin, clopidogrel, oral anticoagulants, passive or active fixation, pacemaker implantation indication, time from implantation, P wave at inclusion, AP ≥ 50%, AP ≥ 75%, VP ≥ 50%, VP ≥ 75%, AP/VP ≥ 50%, AHRE episodes, AF episodes, mortality. All variables with a P value <.2 were entered into a multivariate Cox regression model, where the dependent variable was the development of clinical AF or the appearance of AHRE.
Results are expressed as odds ratio (OR), hazard ratio (HR) and 95% confidence interval (95%CI). All analyses were performed with SPSS 22, (IBM corporation, USA).
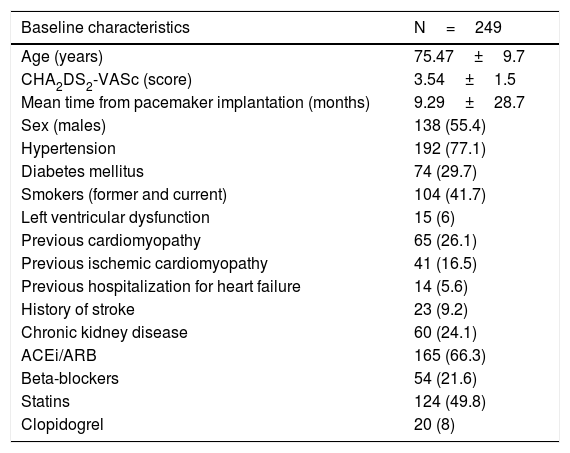
ResultsStudy populationFrom February 2012 to September 2015 we recruited 249 patients, and all patients completed all checkups. 55.4% were men and mean age was 75.47±9.7 years. Mean time from pacemaker implantation was 9.29±28.67 months. Mean CHA2DS2-VASc score was 3.5±1.5, in relation to high rates of hypertension (77.1%), diabetes (29.7%), and previous stroke (9.2%) in the population. Left ventricular dysfunction was present in 6% and 5.6% of participants had been previously hospitalized due to heart failure (Table 1). Twenty-six percent of patients had a previous cardiomyopathy; to note, there were no patients with a diagnosis of infiltrative heart disease. After a mean follow-up of 33.2±11.2 months, 96 patients (38.5%) developed SCAF and 26 patients (10.4%) developed clinical AF. The decision to start anticoagulation when SCAF was detected was individualized and relied on the attending physician.
Basal characteristics.
| Baseline characteristics | N=249 |
|---|---|
| Age (years) | 75.47±9.7 |
| CHA2DS2-VASc (score) | 3.54±1.5 |
| Mean time from pacemaker implantation (months) | 9.29±28.7 |
| Sex (males) | 138 (55.4) |
| Hypertension | 192 (77.1) |
| Diabetes mellitus | 74 (29.7) |
| Smokers (former and current) | 104 (41.7) |
| Left ventricular dysfunction | 15 (6) |
| Previous cardiomyopathy | 65 (26.1) |
| Previous ischemic cardiomyopathy | 41 (16.5) |
| Previous hospitalization for heart failure | 14 (5.6) |
| History of stroke | 23 (9.2) |
| Chronic kidney disease | 60 (24.1) |
| ACEi/ARB | 165 (66.3) |
| Beta-blockers | 54 (21.6) |
| Statins | 124 (49.8) |
| Clopidogrel | 20 (8) |
ACEi, angiotensin converter enzyme inhibitors; ARB, angiotensin receptor blockers.
Data are expressed as no. (%) or mean±standard deviation.
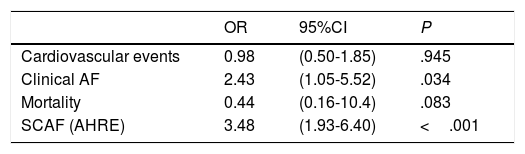
64 patients (25.7%) presented AP ≥ 50%. Sinus node dysfunction was the main reason for pacemaker implantation in patients with AP ≥ 50% (Table 2). In comparison to those with AP <50%, patients with AP ≥ 50% presented a significantly higher risk of developing SCAF (62.5% vs 32.3%; P <.001) and AF (18.7% vs 8.6%; P=.05). In the univariate analysis, patients with AP ≥ 50% had a three-fold risk of developing AHRE (OR, 3.48; 95%CI, 1.93-6.4; P <.01), and a 2-fold risk of developing clinical AF (OR, 2.4; 95%CI, 1.05-5.52; P=.034). The percentage of AP was not related to a higher risk of cardiovascular events or mortality (Table 3). In a multivariate analysis, AP ≥ 50% showed a significant association with the development of SCAF (HR, 2.44; 95%CI, 1.20-4.97; P=.014) (Table 4).
Reasons for pacemaker implantation according to atrial pacing.
| N=249 | AP <50%n=185 | AP> 50%n=64 | P |
|---|---|---|---|
| Sinus node dysfunctionn=49 | 21 (8.4) | 28 (11.2) | <.001 |
| Atrioventricular blockn=130 | 105 (42.2) | 25 (10) | <.001 |
| Conduction abnormalitiesn=70 | 59 (23.7) | 11 (4.4) | <.001 |
AP, atrial pacing.
Data are expressed as no. (%).
Univariate analysis. Atrial pacing ≥ 50% and events.
| OR | 95%CI | P | |
|---|---|---|---|
| Cardiovascular events | 0.98 | (0.50-1.85) | .945 |
| Clinical AF | 2.43 | (1.05-5.52) | .034 |
| Mortality | 0.44 | (0.16-10.4) | .083 |
| SCAF (AHRE) | 3.48 | (1.93-6.40) | <.001 |
95%CI, 95% confidence interval; AF, atrial fibrillation; AHRE, atrial high rate episodes; SCAF, subclinical atrial fibrillation; OR, odds ratio.
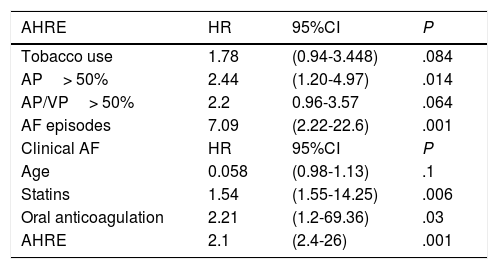
Multivariate analysis. Factors influencing the development of AHRE and clinical AF.
| AHRE | HR | 95%CI | P |
|---|---|---|---|
| Tobacco use | 1.78 | (0.94-3.448) | .084 |
| AP> 50% | 2.44 | (1.20-4.97) | .014 |
| AP/VP> 50% | 2.2 | 0.96-3.57 | .064 |
| AF episodes | 7.09 | (2.22-22.6) | .001 |
| Clinical AF | HR | 95%CI | P |
| Age | 0.058 | (0.98-1.13) | .1 |
| Statins | 1.54 | (1.55-14.25) | .006 |
| Oral anticoagulation | 2.21 | (1.2-69.36) | .03 |
| AHRE | 2.1 | (2.4-26) | .001 |
95%CI, 95% confidence interval; AF, atrial fibrillation; AHRE, atrial high rate episodes; AP, atrial pacing; SCAF, subclinical atrial fibrillation; OR, odds ratio. VP, ventricular pacing.
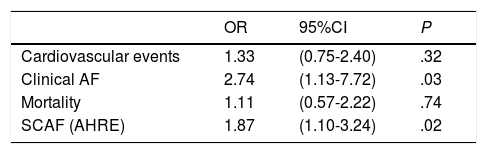
140 patients (56.2%) presented VP ≥ 50%. Atrioventricular block was the main reason for pacemaker implantation in patients showing VP ≥ 50% (Table 5). Patients with VP ≥ 50% presented a significantly higher risk of developing SCAF (46.4% vs 31.6%; P=.03) and AF (25.9% vs 9.7%; P=.051). In the univariate analysis, patients with VP ≥ 50% had a risk almost 2 times higher of developing AHRE (OR, 1.87; 95%CI, 1.10-3.24; P=.02), and almost 3 times higher of developing clinical AF (OR, 2.74; 95%CI, 1.13-7.72; P=.03). No significant differences were found in cardiovascular events or mortality (Table 6). No significant associations were found in the multivariate analysis.
Reasons for pacemaker implantation according to ventricular pacing.
| n=249 | VP <50%n=109 | VP> 50%n=140 | P |
|---|---|---|---|
| Sinus node dysfunction n=49 | 28 (11.2) | 21 (8.4) | <.001 |
| Atrioventricular blockn=130 | 41 (16.5) | 89 (35.7) | <.001 |
| Conduction abnormalitiesn=70 | 40 (16) | 30 (12) | <.001 |
VP, ventricular pacing.
Data are expressed as no. (%).
Univariate analysis. Ventricular pacing ≥ 50% and events.
| OR | 95%CI | P | |
|---|---|---|---|
| Cardiovascular events | 1.33 | (0.75-2.40) | .32 |
| Clinical AF | 2.74 | (1.13-7.72) | .03 |
| Mortality | 1.11 | (0.57-2.22) | .74 |
| SCAF (AHRE) | 1.87 | (1.10-3.24) | .02 |
95%CI, 95% confidence interval; AF, atrial fibrillation; AHRE, atrial high rate episodes; SCAF, subclinical atrial fibrillation; OR, odds ratio.
43 patients (17.3%) presented both AP ≥ 50% and VP ≥ 50% (AP/VP ≥ 50%). In the univariate analysis, no significant associations were found. However, in the multivariate analysis, these patients presented a tendency (although non-statistically significant) towards a higher risk of developing SCAF [HR, 2.2; 95%CI, 0.96-3.57; P=.064] (Table 4). AHRE were shown to be predictors of risk for developing clinical AF [HR, 2.1; 95%CI, 2.4-26; P=.001] (Table 4), as well as AF episodes were predictors of risk for presenting AHRE [HR, 7.09; 95%CI, 2.22-22.6; P=.001] (Table 4).
DiscussionThe aim of our study was to evidence a relationship between the percentage of AP and VP and the incidence of SCAF and clinical AF in patients with dual-chamber pacemakers and no history of AF. We could demonstrate an association between AP and the risk of developing SCAF and clinical AF; moreover, AP was shown to be an independent risk factor. VP was also related to an increased risk of SCAF and clinical AF. Moreover, patients with both AP/VP ≥ 50%, had a tendency towards a higher risk of SCAF. The importance of early detection of SCAF and clinical AF is crucial due to its high embolic risk; the results of our study are of high interest because we could use high percentages of AP and VP as early markers of atrial arrhythmias.
We demonstrated a clear association between AP and the risk of subclinical and clinical AF. Patients with AP ≥ 50% had 3 times more risk of developing AHRE and 2 times more risk of developing clinical AF. Previous studies have related AP with a risk of developing AF, but the mechanisms remain unclear.6 AP produces an atrial conduction delay with intra- and interatrial dyssynchrony, that has been related to the development of AF. The alteration of the conduction times could also be related to higher rates of VP, which has also been associated to an increased risk of AF.8 On the other hand, some studies have suggested AP as a mechanism for AF prevention. The ADOPT study proposed an algorithm that increased the pacing rate when the native rhythm emerged. The result was a reduction in AF risk in patients with sick sinus syndrome.5 In ASSERT, continuous overdrive AP did not prevent AF.9 However, some other studies have shown that AP might be detrimental for new AF onset in patients without sinus node disease, in ICD patients10 and in CRT patients.11 Nevertheless, it remains unclear whether this is related to atrial stimulation or to the increased atrial rate, as in the DAVID trial.12 Therefore, there is great controversy about the relationship between AP and AF, and different mechanisms might be involved. These patients sometimes require AP because of a sick sinus syndrome; therefore, they would have a more diseased atrium, more likely to develop AF. Longer baseline PQ interval has been associated with an increased risk of AF8; this probably also reflects a prolonged atrial conduction time associated with atrial fibrosis. To note, there are different reasons why pacing percentage can be increased. In our study, 22% of patients were under betablockers (Table 1), and the main reason for the pacemaker implantation in patients with AP ≥ 50% was sinus node dysfunction (Table 2). A feasible explanation would be that these patients frequently suffer from tachycardia-bradycardia syndrome and so would be more likely to present AF. In this case, AP could be a marker of risk and not the mechanism for developing AF. On the other hand, 26% of the patients in our study had a previous diagnosis of cardiomyopathy; however, none of them had a diagnosis of cardiac amyloidosis or cardiac sarcoidosis that would predispose them to developing AF.
We could also demonstrate an association between VP and the risk of SCAF and AF. Patients with VP ≥ 50% had almost twice the risk of developing AHRE and almost three times more risk of developing clinical AF. The association between VP and AF has been previously suggested.7 There are some studies that have related VP with an increased risk of permanent AF in patients with sinus node disease.13 However, the effects of VP in paroxysmal AF are controversial. Although previous studies suggested that VP was not detrimental in patients with paroxysmal AF, results of long-MinVPACE conclude that VP was related to an increase in AF burden in patients with paroxysmal AF.14 The mechanisms by which VP will increase the risk for AF are based on the loss of atrioventricular synchrony, which is associated with an increase in atrial pressures, enlargement of the left atrium and mitral regurgitation, all of them related to AF. In this sense, dual-chamber pacing will reduce rates of AF in comparison to VP.13 Also, prolonging programmed atrioventricular intervals with the intention to reduce the rates of VP, has been proposed to reduce the incidence of AF in pacemaker patients.
The percentage of VP can be influenced by the device programming. As already mentioned, it is the protocol of our arrhythmia unit to avoid VP by prolonging the atrioventricular interval and using other dedicated algorithms when necessary. Atrioventricular block was the main reason for pacemaker implantation in patients showing VP ≥ 50% (Table 5). Conventional dual chamber pacing is usually indicated for atrioventricular block, for atrioventricular synchrony maintenance. However, sometimes it can result in high percentage of VP leading to ventricular dyssynchrony, which has been linked by previous studies with an increased risk of AF.13
The role of pacing to prevent AF has been widely discussed. AF is usually the consequence of multiple reentrant wavelets or the impossibility of following a 1:1 activation, leading to a fibrillatory conduction.15 AP would decrease the bradycardic episodes as well as the ectopic beats and so it would eliminate the reentry mechanisms for AF. Multisite AP and different atrial locations of pacing have been tested without showing a decrease in AF risk. AAI or DDDR pacing are less related to the development of AF in comparison to VVI pacing.15 Moreover, atrial rate could have a role in addition to that of AP.
Some studies have suggested an increased risk of hospitalization for heart failure related to ventricular dyssynchrony secondary to VP.16 In our study, we could not demonstrate an association between higher rates of AP and VP and clinical events but pacing was related to a higher incidence of AHRE resembling SCAF, with a previously related higher risk of stroke, as a prior stage for future development of clinical AF. The definition of AHRE has been widely discussed; in our study, the definition of AHRE as episodes ≥ 5minutes was based on studies as MOST4or ASSERT9 that showed that these episodes of> 6 and> 5minutes were related to embolic events. On the other hand, a previous study of our group showed that we should be careful with AHRE interpretation at the early implantation period, since they are more frequent and not related to a higher risk of embolism17. However, in our study, although there was some variability in the times from device implantation, it did not influence the outcomes.
The importance of these results lies on the current increasing interest in the early detection of silent AF, both in patients with cardiac implantable electronic devices, as well as in the global population, through new wearable technologies (iWatch, Kardia…). The detection of episodes of AHRE would be the equivalent of detecting an incipient stage of AF in its progressive course, ranging from paroxysmal AF to persistent and permanent AF. The progression of AF in its different stages has been associated with a greater number of clinical events.18 AHRE have also been related to a high risk of stroke and systemic embolism. Despite the demonstrated increased risk of stroke in patients with SCAF, in the absence of results of randomized controlled trials, the presence of AHRE is not currently an indication for anticoagulation. Two important randomized controlled trials (ARTESIA and NOAH-AFNET) are ongoing, and their results may establish a future indication for the anticoagulation of these patients.19,20.
Study limitationsThe present study has the limitations associated to observational single-center studies. On the other hand, the study includes a real life highly reproducible pacemaker population where an exhaustive evaluation of the pacemaker diagnostic data has been performed, without interfering in other settings of the devices attending to the clinical characteristics of the patients. Only St. Jude Medical devices were included in our study to homogenize the parameters programmed and the detection algorithms that could be different in devices from other companies. On the other hand, our results are translatable to the great majority of pacemaker that use similar detection criteria.
Due to the relatively small population analyzed, our results should be corroborated in future studies. Moreover, the relatively short follow-up period could have been a limitation to detect a possible association between VP and AHRE or clinical AF. As previously mentioned, causality remains unclear and higher percentages of pacing could be markers and not the mechanism for developing AF. However, the results of this study suggesting that pacing increases the risk of SCAF and clinical AF rises the need for future randomized trials that corroborate this association.
ConclusionsPacing is related with a higher risk of developing SCAF and clinical AF in patients with dual-chamber pacemaker and no history of previous AF. Our data suggest that patients presenting a high percentage of AP should be closely followed during routine pacemaker check-ups, assessing for AHRE and clinical AF.
FundingNo funding to declare.
Conflicts of interestNo.
- -
Dual-chamber pacemakers provide reliable detection of AHRE.
- -
AHRE have been related to a higher risk of clinical AF and thromboembolic events.
- -
AP is an independent predictor for developing AHRE and AF.
- -
VP is related with a higher incidence of AHRE and AF.
- -
These risk markers are important since the risk of stroke is increased in these patients.
- -
A close follow-up of these patients could help in obtaining an early diagnosis of AF.