The epicardial adipose tissue (EAT) is a metabolically active fat depot that has shown to be implicated in the development of coronary artery disease (CAD) and the prediction of cardiovascular events. However, these findings might be influenced by body size since previous studies have demonstrated a strong correlation between the amount of epicardial fat and anthropometric measurements. Our objective was to assess the association between indexed-EAT (EAT-i) volume and the presence of CAD and cardiovascular events in a cohort of symptomatic patients.
MethodsPatients referred for cardiac computed tomography from March 2010 to April 2017 were included. CAD was defined as the presence of obstructive coronary stenosis or a high amount of coronary calcium. EAT was assessed using a semiautomatic software and indexed to body surface area. A regression analysis was performed to determine the relationship between relevant CAD and EAT-i, age, sex, cardiovascular risk factors, and comorbidities. The association between EAT-i and cardiovascular events was analyzed with Cox regression models.
ResultsA total of 179 patients were recruited (56±12 years, 57.5% male). EAT-i (P=.003), age (P<.001) and male sex (P<.001) were significantly and independently associated with CAD. During follow-up (5.5 years, 2.4–6.4) patients with elevated EAT-i showed an increased risk of events (HR, 2.44; 95%CI, 1.07–5.56; P=.033).
ConclusionsIn symptomatic patients, an increased volume of indexed epicardial fat was associated with obstructive CAD as well as with cardiovascular events.
El tejido adiposo epicárdico (TAE) es un depósito metabólicamente activo que ha demostrado estar implicado en el desarrollo de enfermedad arterial coronaria (EAC) y en la predicción de eventos cardiovasculares. Sin embargo, estos hallazgos podrían estar influidos por el tamaño corporal, ya que existe correlación entre la cantidad de grasa epicárdica y las medidas antropométricas. Nuestro objetivo fue analizar la asociación entre el TAE indexado (TAE-i) con la EAC y los eventos en una cohorte de pacientes sintomáticos.
MétodosSe incluyeron pacientes derivados para la realización de tomografía computarizada coronaria entre marzo de 2010 y abril de 2017. Se definió EAC por la presencia de estenosis coronarias obstructivas o una cantidad elevada de calcio coronario. Se cuantificó el TAE mediante un programa informático semiautomático y se indexó el volumen al área de superficie corporal. Se realizó un análisis de regresión para determinar la relación entre la presencia de EAC y TAE-i, edad, género, factores de riesgo cardiovascular y comorbilidades. La asociación entre TAE-i y eventos se analizó con modelos de regresión de Cox.
ResultadosSe incluyeron 179 pacientes (56±12 años, 57,5% varones). El TAE-i (p=0,003), la edad p<0,001) y el sexo masculino (p<0,001) se asociaron de forma independiente con la presencia de EAC. Los pacientes con mayor TAE-i mostraron un mayor riesgo de eventos cardiovasculares durante el seguimiento (5,5 años, 2,4-6,4) (HR=2,44; IC95%, 1,07-5,56; p=0,033).
ConclusionesEn pacientes sintomáticos un mayor volumen de TAE-i se asocia con EAC obstructiva, así como con eventos cardiovasculares.
Cardiovascular disease is still a leading cause of morbidity and mortality, in spite of improvements in outcomes,1 and the prevalence of some risk factors, particularly diabetes or obesity, is increasing.2 Furthermore, it has been suggested that fat distribution is associated with the development of coronary artery disease (CAD), independently of the global amount of fat.3 Epicardial adipose tissue (EAT) is a part of the visceral fat located between the myocardium and the pericardium,4 it surrounds the heart while embracing the coronary arteries.5 The EAT is a metabolically active fat depot, since it is implicated in the development of CAD due to its close anatomical relationship with the coronary arteries, eventually related with a mechanism of paracrine and vasocrine secretion of proinflammatory cytokines4,6 which could trigger the atherosclerotic process. Cardiac computed tomography (cardiac CT) allows a simultaneous assessment of coronary atherosclerotic plaque and EAT volume,7 and it is the preferred imaging modality for EAT quantification because of its high spatial resolution, accurate assessment, and wide availability.8
Prior studies have already shown the relationship between EAT, CAD and cardiovascular events.3,5,7–13 However, the association might be confused since there is a strong correlation between the amount of epicardial fat and anthropometric measurements14–17; in consequence, the adjustment of the amount of EAT by body size seems crucial. So far only a few studies have reported the relationship after adjustment by body size.18–22
Furthermore, in our study we recruited patients with chest pain of potential cardiac origin, which is at odds with the previously mentioned studies, which included asymptomatic patients20 or patients with unstable angina or known CAD.19,22
The purpose of our study was to investigate the association between indexed-EAT (EAT-i) volume and cardiovascular risk factors, the presence of CAD and cardiovascular events in a cohort of symptomatic patients with chest pain or dyspnea of unknown origin.
MethodsStudy populationDuring the period from March 2010 to April 2017, we retrospectively recruited consecutive patients with chest pain or dyspnea of unknown origin, attended at the cardiology outpatient clinic in 2 University Referral Hospitals in our area, who underwent a cardiac CT performed for clinical reasons according to current guidelines,23 composing a retrospective cohorts study. Subjects were excluded if they had any other cardiomyopathy or if the cardiac CT study was unsatisfactory, owing to low-quality image acquisition. The study protocol was approved by our local ethics committees. The study was exempted from the need of an informed consent by our local ethics committee given the retrospective character of our study and the long-term recruitment. Anonymity of the database was ensured at all times.
Imaging protocol of cardiac computed tomographyA Somaton 64-slice multi-detector CT (Siemens Healthineers, Germany) was used for scanning. Breath holding was utilized to minimize motion artefact. Oral and/or intravenous betablockers were administered as needed to lower heart rate below 65 beats per minute to minimize reconstruction artefacts. Thereafter, a 0.4mg sublingual dose of nitroglicerin was given. A total dose of 1mg/kg intravenous iodine 350mg/g contrast was given to obtain a noninvasive coronary angiography when appropriate. Patients showing high calcium scores (> 400 Agatston Units [AU]) did not receive contrast, and a non-contrast study including only coronary artery calcium score (Agatston score) was performed. The cardiac CT was performed according to the following protocol: tube voltage 100kVp, tube current 55mAs, pitch 0.31, gantry rotation time 0.3s, and collimation 128×0.6mm. The images were reconstructed with a slice thickness of 3 and 0.8mm for calcium scoring and coronary angiography, respectively. The coronary calcium score was calculated according to the method described by Agatston et al.24 Scans were analyzed by an experienced radiologist using a 3-dimensional workstation. We defined CAD as the presence of obstructive coronary stenosis (at least one coronary stenosis > 50%) or a high amount of coronary calcium (Agatston Score≥400AU), based on a definition previously accepted in a few studies.9,13
Quantification of epicardial adipose tissueEAT was identified on non-contrast data sets as a hypodense layer surrounding the myocardium and limited by the pericardium. A volumetric software was used to integrate the whole set of user-defined area traces, from the most cranial at the level of the pulmonary artery to the most caudal at the level of the base of the heart that seemingly included any pericardial content, to come up with an overall EAT volume (cm3).
OutcomesAll subjects were followed for adverse cardiovascular events, including cardiac death, myocardial infarction, revascularization (occurring more than 30 days after the CT), new-onset atrial fibrillation, acute ischemic stroke, transient ischemic attack, or cardiac hospitalizations. Follow-up information was obtained by review of the corresponding medical records in our electronic hospital information system, or telephone contact if needed.
Statistical analysisContinuous variables were expressed as means±standard deviations, or medians with [interquartile range], and were compared with Student t test. Categorical variables were reported as frequencies and percentages and compared using Fisher's exact test or the Chi-square test, according to the data size. A Kolmogorov–Smirnov test was firstly used to check the assumption of normal distribution. Taking into consideration the anthropometric variability of EAT volume, we also made the statistical analysis after having indexed EAT to body surface area (EAT-i). A binary logistic regression analysis was performed to determine the relationship between relevant CAD and EAT-i, age, gender, cardiovascular risk factors and cardiovascular comorbidities. Cardiovascular risk factors (hypertension, hyperlipidemia, diabetes mellitus, smoking and obesity) and comorbidities (chronic kidney disease, cerebrovascular disease, peripheral arterial disease, and atrial fibrillation) were grouped in 2 different variables (ie, “cardiovascular risk factors” and “comorbidities”). Odds ratio and 95% confidence interval (95%CI) were calculated for each covariate, as well as the calibration of the model by using Hosmer–Lemeshow test, and C-statistic to assess discrimination. The association of EAT with cardiovascular events during follow up was assessed using Cox regression, in a model which included median EAT-i, age, cardiovascular risk factors, and comorbidities. The first-degree interaction between chronic coronary syndrome and EAT-i was studied using likelihood ratio statistics and the backward elimination method (chunk test). For survival analysis, Kaplan–Meier curves were created to estimate the distribution of cardiovascular events, depending on the presence of elevated EAT-i; the log-rank test was used to test differences in survival between groups. A random subset of 10 patients was selected to evaluate reproducibility of EAT volume measurements, and it was assessed using intra-class correlation coefficient. A 2-tailed probability value of P<.05 was considered statistically significant. Statistical analysis was performed using SPSS software (IBM SPSS statistics, version 22.0 for Mac, Armonk, United States).
ResultsBaseline characteristicsPatient baseline characteristics are shown in Table 1. A total of 179 subjects were included in our analysis, with a mean age of 56±12 years, 103 (57.5%) were men, with a median of 2 [range 1–3] cardiovascular risk factors per individual. In our study ninety-one subjects showed no plaque (50.8%), 35 (19.6%) non-obstructive plaques, and 16 (9%) subjects showed at least one coronary stenosis > 50% (obstructive plaques). A total of 53 patients (29.6%) were diagnosed of having CAD. One-vessel disease was demonstrated in 10 subjects (5.6%), 2-vessel disease in 5 subjects, and three-vessel disease in 1 subject. Left main disease was demonstrated in one patient. Median Agatston Score was 10UA [range 0–380UA]. Absence of coronary calcium was reported in 79 (44.4%) individuals, while an Agatston Score above 400 was found in 42 (23.5%).
Baseline Characteristics of the total cohort and stratified by cardiovascular events during follow-up.
| Total cohort (n=179) | Cardiovascular events | P | ||
|---|---|---|---|---|
| No (n=142, 79.3%) | Yes (n=37, 20.7%) | |||
| Age, years | 55.66±12.06 | 54.18±11.58 | 61.35±12.35 | .001 |
| Male sex, n (%) | 103 (57.5) | 77 (54.4) | 26 (70.3) | .079 |
| Cardiovascular risk profile | – | – | – | - |
| Hypertension, n (%) | 103 (57.5) | 76 (53.5) | 27 (73.0) | .033 |
| Type 2 diabetes mellitus, n (%) | 35 (19.6) | 22 (15.5) | 13 (35.1) | .007 |
| Dyslipidaemia, n (%) | 107 (59.8) | 84 (59.2) | 23 (62.2) | .740 |
| Smoking history, n (%) | 79 (44.1) | 59 (41.5) | 20 (54.1) | .172 |
| Body mass index, kg/m2 | 28.04 [25.34–31.83] | 28.60±4.94 | 29.55±4.50 | .291 |
| Total cholesterol, mg/dl | 188 [152–214] | 187±43 | 184±70 | .679 |
| LDL-cholesterol, mg/dL | 105 [85–133] | 110±37 | 103±35 | .252 |
| Triglycerides, mg/dL | 112 [80–167] | 130±76 | 152±96 | .140 |
| HDL-cholesterol, mg/dL | 47 [40–57] | 50±14 | 44±12 | .008 |
| Family history of CAD, n (%) | 19 (10.6) | 18 (12.7) | 1 (2.7) | .079 |
| Cerebrovascular disease, n (%) | 7 (3.9) | 4 (2.8) | 3 (8.1) | .139 |
| Peripheral arterial disease, n (%) | 9 (5.0) | 3 (2.1) | 6 (16.2) | <.001 |
| Chronic coronary syndrome, n (%) | 14 (7.8) | 9 (6.3) | 5 (13.5) | .148 |
| Atrial fibrillation, n (%) | 14 (7.8) | 8 (7.9) | 6 (21.4) | .042 |
| Chronic kidney disease, n (%) | 7 (3.9) | 3 (2.1) | 4 (10.8) | .015 |
| Aspirin, n (%) | 58 (32.4) | 41 (28.9) | 17 (45.9) | .048 |
| Beta-blockers, n (%) | 50 (27.9) | 40 (28.2) | 10 (27.0) | .866 |
| Statins, n (%) | 76 (42.5) | 57 (40.1) | 19 (51.4) | .219 |
| ACE inhibitors/AT II blockers, n (%) | 79 (44.1) | 59 (41.5) | 20 (54.1) | .172 |
| Coronary calcium score, AU | 10 [0–380] | 228.09±609.56 | 775.92±1000.91 | .003 |
| EAT volume, cm3 | 96.67 [72.36–131.24] | 100.10±43.44 | 121.64±40.44 | .007 |
| Indexed EAT volume, cm3/m2 | 52.51 [40.78–68.15] | 53.78±20.93 | 63.79±19.22 | .009 |
ACE indicates angiotensin-converting enzyme; AT-II, angiotensin II receptor blocker; AU, Agatston Units; CAD, coronary artery disease; EAT, epicardial adipose tissue; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Results are shown as mean±standard deviation, n (%) or median [interquartile range].
Median EAT volume was 96.67cm3 [range 72.36–131.24cm3], and median EAT-i volume 52.51cm3/m2 [range 40.78–68.15cm3/m2]. Patients with type 2 diabetes mellitus (67.56cm3/m2 vs 53.01cm3/m2; P<.001), hyperlipidemia (59.12cm3/m2 vs 50.98cm3/m2; P=.011) and hypertension (60.81cm3/m2 vs 49.13cm3/m2; P<.001) had a significantly higher EAT-i volume. EAT-i volume was also greater in patients with CAD (68.29cm3/m2 vs 50.62cm3/m2; P<.001).
Cardiovascular risk factors and the presence of coronary artery diseaseCompared with subjects without CAD, patients with CAD were significantly older (P<.001), were men in a greater proportion (P<.001) and more likely had cardiovascular risk factors: namely hypertension (P<.001), diabetes (P<.001), and a greater mean body mass index (P=.002).
Associations between epicardial adipose tissue and coronary artery diseaseLogistic regression analysis showed that EAT-i (OR, 2.6 per increase in 1cm3/m2 (95%CI, 1.13–6), P=.003), age (P<.001) and male sex (P<.001) were significantly and independently associated with CAD, in a well calibrated model that further showed a good discrimination (Hosmer–Lemeshow test Chi-square=2.039, P=.980; C-Statistic AUC 0.845, 95%CI, 0.784–0.907, P<.001), which included also cardiovascular risk factors and comorbidities.
Epicardial adipose tissue and cardiovascular outcomesDuring a median of 5.53 years follow-up period [range 2.37–6.37 years], 37 cardiovascular events occurred: 9 acute coronary syndromes, 13 late revascularizations, 10 cardiovascular hospitalizations (5 due to acute decompensated heart failure, 2 myopericarditis, 1 third degree heart block, and 2 chest pains of unknown origin), 4 strokes or transient ischemic attack, and one cardiovascular death. Patients with cardiovascular events (Table 1) showed higher EAT-i volume than patients without cardiovascular events (63.79±19.22 vs 53.78±20.93cm3/m2, P=.009). Moreover, they were significantly older (61.35±12.35 years vs 54.18±11.58 years, P<.001), with a higher proportion of hypertension (73.0% vs 53.5%, P=.033) and type 2 diabetes mellitus (35.1% vs 15.5%, P=.007).
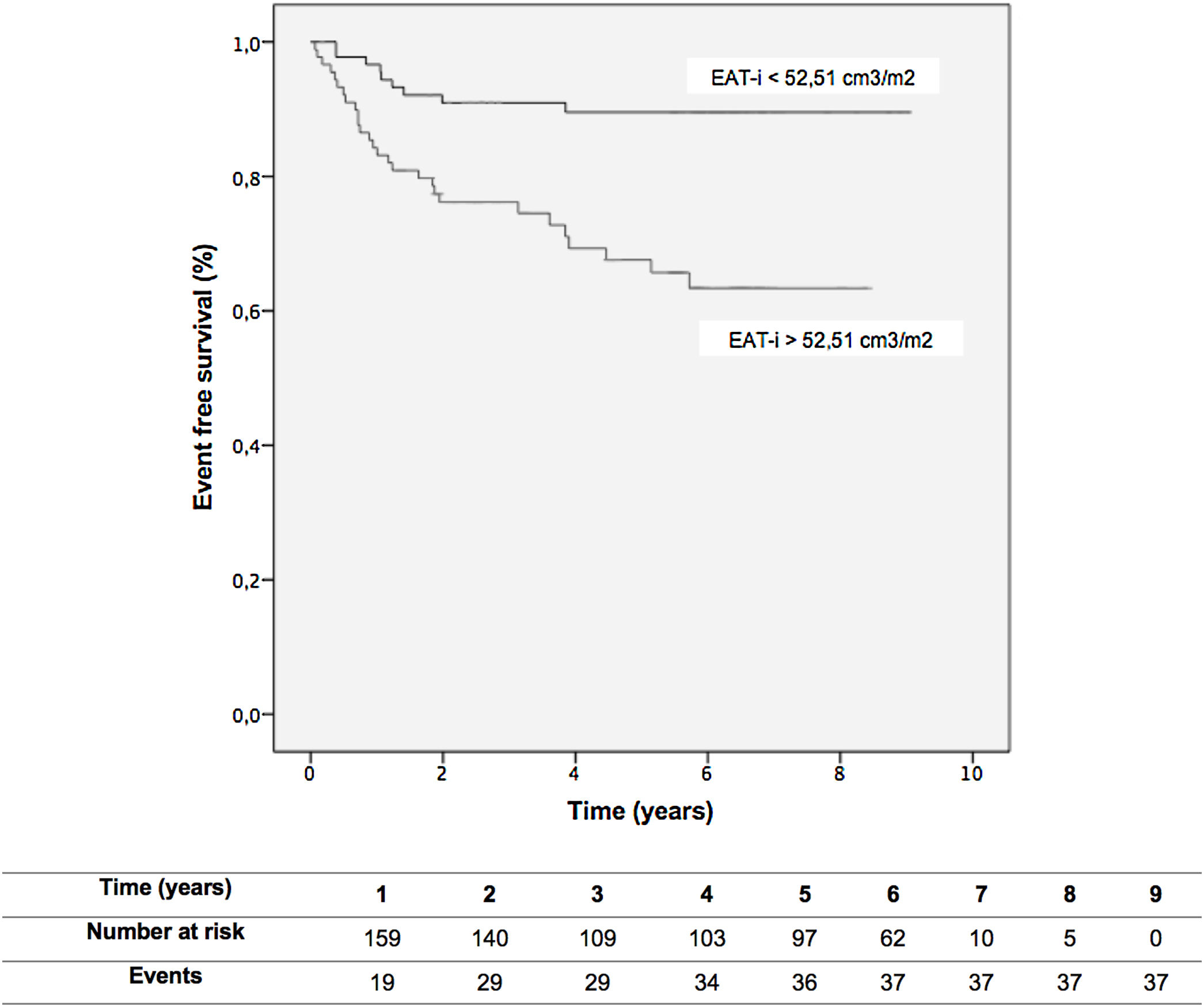
A Kaplan–Meier survival analysis is shown in Fig. 1. Those patients with elevated EAT-i showed a significantly higher risk of cardiovascular events during follow-up (log-rank test P<.001, Fig. 1). The selected cutoff value was the median EAT-i (52.51cm3/m2, negative predictive value 90%, positive predictive value 31%). Notably, using multivariable adjusted Cox regression analysis, median EAT-i showed an independent association with adverse cardiovascular events (hazard ratio, 2.44; 95%CI, 1.07–5.56; P=.033) (Table 2). No significant interaction was found between previous chronic coronary syndrome and EAT-i (P=.961).
Cox regression for predictors of cardiovascular events.
| Variable | HR | 95%CI | P |
|---|---|---|---|
| Age, years | 13.82 | 0.23–837.99 | .210 |
| Median EAT-i* | 2.44 | 1.07–5.56 | .033 |
| Cardiovascular risk factors | 1.17 | 0.90–1.54 | .246 |
| Comorbidities | 1.52 | 1.05–2.21 | .026 |
95%CI, 95% confidence interval; EAT-i, epicardial adipose tissue indexed to body surface area; HR, hazard ratio.
“Cardiovascular risk factors” includes hypertension, diabetes mellitus, hyperlipidemia, smoking and obesity; “Comorbidities” includes chronic kidney disease, cerebrovascular disease, peripheric arteriopathy an atrial fibrillation.
The intra-observer reproducibility calculated from a subset of 10 random patients was excellent with an intra-class correlation coefficient value of 0.976 (95%CI, 0.908–0.994).
DiscussionIn the present study we have shown a significant and independent association of elevated EAT volume indexed to body surface area, with the presence of CAD and cardiovascular events, irrespective of traditional cardiovascular risk factors and comorbidities.
EAT is a metabolically active annular fat depot, that has been implicated in the pathogenesis of coronary atherosclerosis. This concept was strengthened by the observations of intramyocardial coronary segments that were free of atherosclerotic disease.4 Numerous investigations have already described a significant association of EAT volume with atherogenic risk factors.7,14,25 These observations are in line with our findings, as we observed a greater amount of epicardial fat in individuals with cardiovascular risk factors. However, the question arises whether epicardial fat is just a marker of visceral adiposity acting as a para-phenomenon and signaling the presence of other cardiovascular risk factors, or a causality relationship can be established.26
Some authors failed to show an association between epicardial fat and CAD. Chaowalit et al. published a study of consecutive patients who underwent echocardiography and invasive coronary angiography, and they found no correlation between EAT and angiographic lesions.27 Bettencourt et al. could not report a significant independent relation of EAT volume with the severity of CAD either, speculating that EAT might have a role only in the initial phase of the coronary atherosclerotic development process.28 Along the same lines, in a more recent study in the setting of symptomatic individuals, EAT was found as a predictor of coronary calcium but no predictive value in the burden of CAD could be established, and the authors hypothesize that the lack of relation could be due to the low prevalence of CAD in their sample.29
However, in our study, EAT-i was independently associated with CAD, after adjustments for comorbidities and cardiovascular risk factors. It should be stressed that the prevalence of CAD in our sample was similar to that found in other series of symptomatic patients in which a relationship between EAT and CAD was being investigated.4,7,9,11,29–31 Accordingly, a large number of prospective studies have already demonstrated a relationship between epicardial fat and CAD. In a cohort of asymptomatic patients, Goeller et al. investigated whether EAT was related to early atherosclerosis, finding that indeed EAT volume was higher in subjects with early and more advanced atherosclerosis.12 Also dealing with asymptomatic patients, Eisenberg et al. showed an independent predictive prognostic value of EAT in new and fully automated approach based on deep-learning techniques.5 On the other hand, in patients with stable chest pain, Zhou et al. came to the conclusion that EAT significantly improved prediction of obstructive CAD above cardiovascular risk factors and coronary calcium in Chinese Population.9 In the PROMISE trial, a symptomatic cohort with increased cardiovascular risk, authors showed a robust relationship between EAT and extension of CAD, even though they did not find association between EAT and adverse cardiac events.10 Recently, a meta-analysis published by Mancio et al. with approximately 20000 low-risk subjects, found that EAT is linked to CAD, coronary events and myocardial ischemia, and these associations remained significant after adjusting for cardiovascular risk factors.13 Nevertheless, only a few studies attempted to normalize the EAT volume for body surface area,18,20–22 although none of them demonstrated a significant independent association of EAT with CAD in low-risk symptomatic patients, as we did in our study.
LimitationsOur study has some limitations that should be acknowledged, mainly related to the retrospective nature of the study as well as its cross-sectional design, which precludes obtaining conclusions that could imply a cause–effect relationship. Besides, the sample size and the scarce number events observed make our observations difficult to interpret, requiring additional large-scale studies to expand and understand the results. Nevertheless, despite the relatively small sample size, the baseline characteristics of our population were in line with bigger studies.5
Our results contribute to an expanding body of evidence for the independent role of EAT volume in the development of CAD and cardiovascular events, and it supports the idea of EAT as a tool which could enhance cardiovascular risk stratification.
ConclusionsIn the setting of symptomatic patients with chest pain or dyspnea of unknown origin, an increased volume of epicardial fat was associated with obstructive CAD as well as with cardiovascular events.
Epicardial adipose tissue, a metabolically active depot, is associated with CAD and adverse cardiac events. However, these findings might be confounded by body size since there is a strong correlation between the amount of epicardial fat and anthropometric measurements. Thus far, this relationship after adjustment by body size has not been properly studied.
Does it contribute anything new?Our results showed a significant and independent association between elevated EAT volume indexed and body surface area with the presence of CAD and cardiovascular events, irrespective of traditional cardiovascular risk factors and comorbidities.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors’ contributionsThe work includes only authors who have made an important contribution to the study and can warrant their involvement. All authors are responsible for the content and have read and approved the manuscript.
Conflicts of interestL. Consuegra-Sánchez is associate editor of REC: CardioClinics. The journal's editorial procedure to ensure impartial handling of the manuscript has been followed. The rest of authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.